- Login
- Search
- Contact Us
-
Have a question? Our team is here to help guide you on your automation journey.
-
Explore support plans designed to match your business requirements.
-
How can we help you?
-
- AI
AI Without the Hype From pilot to full deployment, our experts partner with you to ensure real, repeatable results. Get Started
- Automation Anywhere AI
-
- Solutions
Featured Agentic Solutions
Accounts Payable Invoice automation—No setup. No code. Just results. Accounts Payable
Customer Onboarding Scale KYC/AML workflows. Customer Onboarding
Customer Support Keep queues moving, even at peak load. Customer Support
Healthcare RCM Revenue cycle management that runs itself. Healthcare RCM
- Products
Platform Features
- Agentic process automation (APA)
- Robotic Process Automation (RPA)
- View all Products
-
- Resources
Get Community Edition: Start automating instantly with FREE access to full-featured automation with Cloud Community Edition.
Featured
 Named a 2025 Gartner® Magic Quadrant™ Leader for RPA.Recognized as a Leader for the Seventh Year in a Row Download report Download report
Named a 2025 Gartner® Magic Quadrant™ Leader for RPA.Recognized as a Leader for the Seventh Year in a Row Download report Download report- Become an Expert
- Developer Tools
- Get Support
- View all resources
-
- Partners
Find an Automation Anywhere Partner Explore our global network of trusted partners to support your Automation journey Find a Partner Find a Partner
- Find a Partner
- For Partners
-
- Imagine 2026
Event
Get ready for Imagine 2026
From agentic AI to end‑to‑end automation, be a part of the flagship event where our community gathers to build, learn, and lead. Register today
Countdown
Blog
Identifying Automation Opportunities for Life Sciences
The Everest Group has identified processes where life sciences can reap the most benefits from intelligent automation.

Life sciences companies began the year 2020 with significant challenges, including COVID-19. The virus created an instant demand to fast-track products without sacrificing quality and profitability and to ensure continuity of medical supplies. Add to that challenge the increasing pressure on life sciences companies from generics. Development and manufacturing costs were increasing, along with customer expectations. And there were regulatory changes.
For many industry leaders, intelligent automation—a combination of Robotic Process Automation (RPA) and artificial intelligence—has helped them meet the challenges by transforming their operations to increase ROI, efficiency, quality, and compliance and supporting the accelerated push of the drug cycle from lab to market.
Examining the challenges and the solution
In its report, “Enterprise Value Chain Approach (EVCA) to Identify Automation Opportunities in Life Sciences,” the Everest Group examines life sciences challenges, how intelligent automation can help companies overcome them, and which life sciences processes are the best candidates for automation.
The four quadrants of automation opportunities
The Everest Group diagram below summarizes the business processes within life sciences. 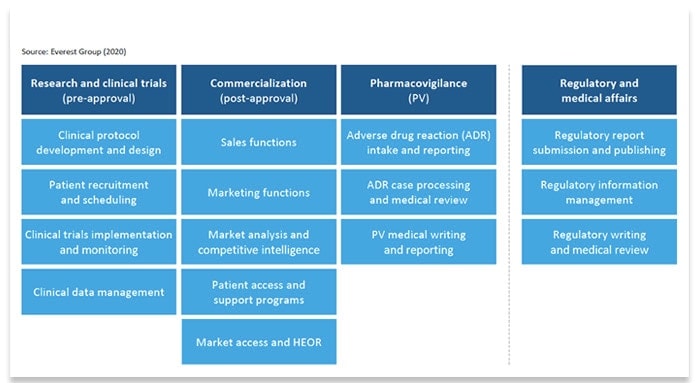
The Everest Group evaluated all of these processes using a proprietary methodology to assess the suitability of processes for automation. Named the “Enterprise Value Chain Approach” (EVCA), this five-step methodology helps businesses identify business processes, analyze each process, spotlight high-value opportunities for automation, and define metrics to prioritize those opportunities.
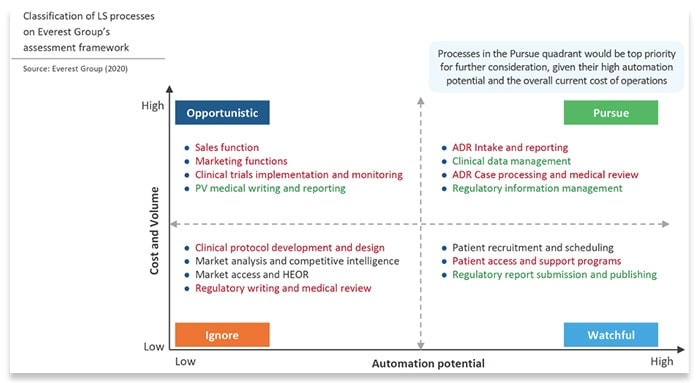
Using EVCA scores, processes are divided into the following four quadrants based on cost savings potential and overall automation potential:
- Pursue: Processes that score high on cost savings potential and automation potential fall into this quadrant. These are the highest priority processes to automate.
- Opportunistic: Processes score high on cost savings, but low on automation potential, should be automated only on an “opportunistic” basis. Such opportunities should be pursued only when a use case presents itself during the regular course of business operations where the benefits outweigh the costs.
- Watchful: The processes in this quadrant return relatively little value but are easy to automate. These lower-priority processes can be automated when budget/resource capacity allows for it.
- Ignore: These are difficult to automate and return very little value. Ignore them.
Based on Automation Anywhere research, we’ve identified four top life sciences use cases for automation. They are:
- Pharmacovigilance (PV) and complaint handling, including call centers for case management
- Supply chain management, which includes managing vendors and tracking products through the channel
- Clinical trial management for participant recruiting, patient monitoring, and regulatory documentation
- Commercial operations, sales, and marketing
The Everest Group also considers pharmacovigilance to be a top use case for automation. The company has placed most of the processes listed in its “pursue” automation quadrant—such as alternative dispute resolution (ADR) intake and reporting and ADR case processing—under the umbrella of pharmacovigilance.
The challenges of pharmacovigilance
If nothing else, PV is about information: clinical trials, post-market surveillance, adverse event cases, and other types of data. With legacy systems, the process of gathering and analyzing that data—structured and unstructured—has been a labor- and time-intensive, manual task. And that can make pharmacovigilance a weak link in the life sciences business chain.
A manual system can slow down processing when the accelerated delivery of vaccines and therapies is essential not just for competitive advantage but for beating the clock to save lives. Manual processing can also be costly as the volume of data increases—requiring additional resources—such as in the current rush to find a vaccine for COVID-19.
Manual systems can lead to informational silos that can make it difficult to get an accurate view of a company’s PV landscape. They can lead to human errors with the potential to affect many areas of the business chain, including product development and quality as well as compliance. And they can keep employees from focusing on higher-value work to enhance a company’s efficiency and productivity.
The benefits of intelligent automation for pharmacovigilance
Intelligent automation (IA), with the use of software bots, can automate, streamline, and accelerate complex, highly regulated life sciences processes. In terms of PV, it can reduce the time and effort spent on data entry—tracking safety issues, optimizing customer support, managing communications, and reporting—and increase the time for human workers to pursue higher-level work.
Bots can:
- Handle all types of data, gathering, extracting, and analyzing unstructured and structured information from a wide variety of sources—with no human intervention
- Eliminate bottlenecks by scaling to meet demand as data volume increases and by processing data around the clock for continuous operation
- Offer greater collaboration and visibility, eliminating informational and process silos to enhance process visibility and allow for real-time information sharing. They can not only monitor processes but also communication channels such as emails.
- Increase accuracy by eliminating human error and reduce case costs by increasing efficiency
See how intelligent automation can help improve the health of your life sciences business.
Start and Scale Automation with the Free Everest Report
Stay up to date:
Catherine Calarco, vice president of innovation evangelism at Automation Anywhere, has more than 20 years of global leadership in life sciences, digital medicine, and technology. She's known for driving significant B2C/B2B revenue growth and developing innovative, award-winning digital health

For Students & Developers
Start automating instantly with FREE access to full-featured automation with Cloud Community Edition.

